М (Al(OH)₃) =27 + 16*3 + 3 = 78
ω (Al) =
ω (О) =
ω (Н) =
Al(OH)3
Mr(Al(OH)3) = 27 + 3*16 + 1*3 = 78г
w(Al) = Ar(Al)*n(Al)/Mr(Al(OH)3) = 27*1/78 = 0,35 або 35%
w(O) = 16*3/78 = 0,62 або 62%
w(H) = 1*3/78 = 0,03 або 3%
М (Al(OH)₃) =27 + 16*3 + 3 = 78
ω (Al) =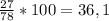
ω (О) =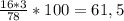
ω (Н) =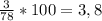
Al(OH)3
Mr(Al(OH)3) = 27 + 3*16 + 1*3 = 78г
w(Al) = Ar(Al)*n(Al)/Mr(Al(OH)3) = 27*1/78 = 0,35 або 35%
w(O) = 16*3/78 = 0,62 або 62%
w(H) = 1*3/78 = 0,03 або 3%